CHEM 1001/CHM1020/CHM1025C/CHM1032C Name: __Answers___
Module Three Sample Pretest
Module
Three: Part D Electron Dot
Formulas 10 points
Using
the periodic chart, draw the electron dot formulas of the following elements (the
numbers shown are the element’s atomic number and mass number):
|
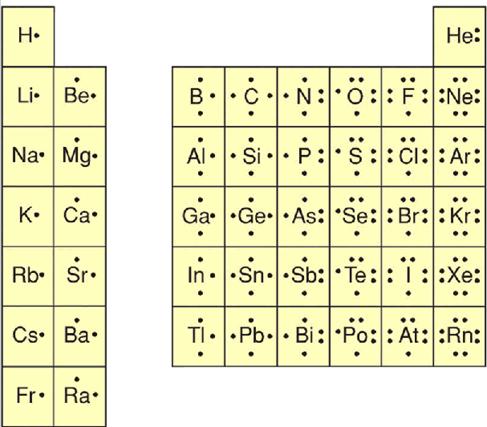
1. 6C12 2. 14Si28 3. 9F19 4. 11Na23 5. 15P31 6. 1H1 7. 7N14 8. 8O16 9. 10Ne20 10. 16S32 |
Complete Table of
Representative Elements’ Dot Structures |
Module
Three: Part F Periodic Ionic Properties 10 points
Using
a periodic chart, write the ionic character (monoatomic
ionic charge) of the following elements: (The number before the element is its atomic
number)
1. 19 K _1+_____ 6.
9F __1-__
2. 20Ca _2+____ 7. 1H __1+__ __1-___
3. 7N _3-____ 8. 16S __2-__
4. 17Cl _1-____ 9. 10Ne __0___
5. 53I _1-____ 10. 15P __3-___