CHEM 1001/CHM 1020/ Chm1025C/CHM1032C Name:________
Module 6 Sample Pretest
Part
F Volume-Volume Stoichiometry
5 points
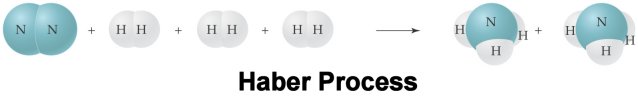
In the Haber process, nitrogen
N2 and hydrogen H2 gases combine to give ammonia
gas NH3 as the only product. If 5.55 L of nitrogen gas
completely reacts, calculate the volume of ammonia that is produced. Assume all
volumes of gas are measured under constant conditions of 500 oC and 300 atm
pressure. How many liters of hydrogen are required to complete react the 5.55L
of nitrogen?
 Using ChemiCalc
to balance the equation.
Using ChemiCalc
to balance the equation.
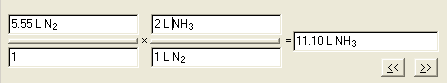
Step
#1 Write the balanced reaction
N2 + 3 H2 à 2 NH3
Step
#2 Write the question in Dimensional Analysis format:
__?__ L N2 = 5.55
L NH3
Step
#3 Setup the dimensional analysis problem.
Think
about the Road map of Stoichiometry:
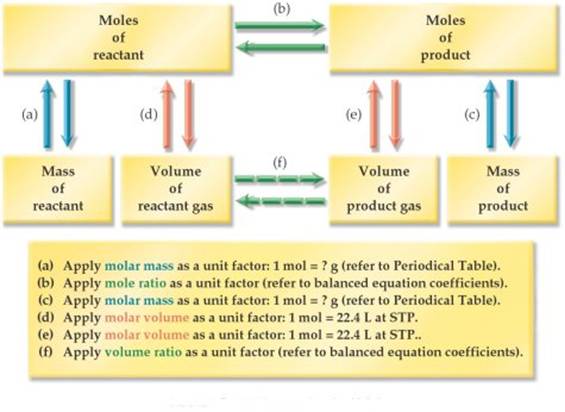
It
is one step with a direct volume to volume unit factor similar to the mole
ratio. See (f) above.
See Section 10.6 of Chapter 10 as this
is the very problem from your textbook on page 264.
First answer is 11.1 L
NH3
Second
Question: __?__ L H2 = 5.55
L NH3

Second answer: 16.65 L H2
Part
G Mass-Volume Stoichiometry 5 points
Potassium chlorate is
used in the lab to make oxygen gas by the following (unbalanced) reaction:
KClO3 à
KCl
+ O2
How liters of
oxygen at STP may be made from reacting 1.226 grams of Potassium chlorate?
Step #1: Write and
Balance the Equation:
2 KClO3 à 2 KCl +
3 O2
Step#2
Write
the question in Dimensional Analysis format:
__?__ L O2 = 1.226
g KClO3
Step#3
Think
about the Stoichiometry Road Map
a. Change grams of KCLO3
to moles KCLO3 using molar mass of KCLO3
b. Change moles of KCLO3 to moles O2 using the mole
ratio from the balanced equation.
c. Change moles of O2 to Liters of O2 using the
molar gas volume constant assuming STP.
Step #4 Setup and Solve the Dimensional Analysis
Sequence:

Your answer is 0.336 L O2